Therefore, a hydrogen molecules mass is 2 amu. At room temperature, hydrogen is stable and unreactive. A hydrogen, nitrogen, or oxygen molecule, consists of two identical atoms of each of those respective elements. Hydrogen is more soluble in organic solvents and less soluble in water. Chemical Characteristics Hydrogen is a highly (combustible) flammable gas. Neutron number plus atomic number equals atomic mass number: N ZA. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol N. 
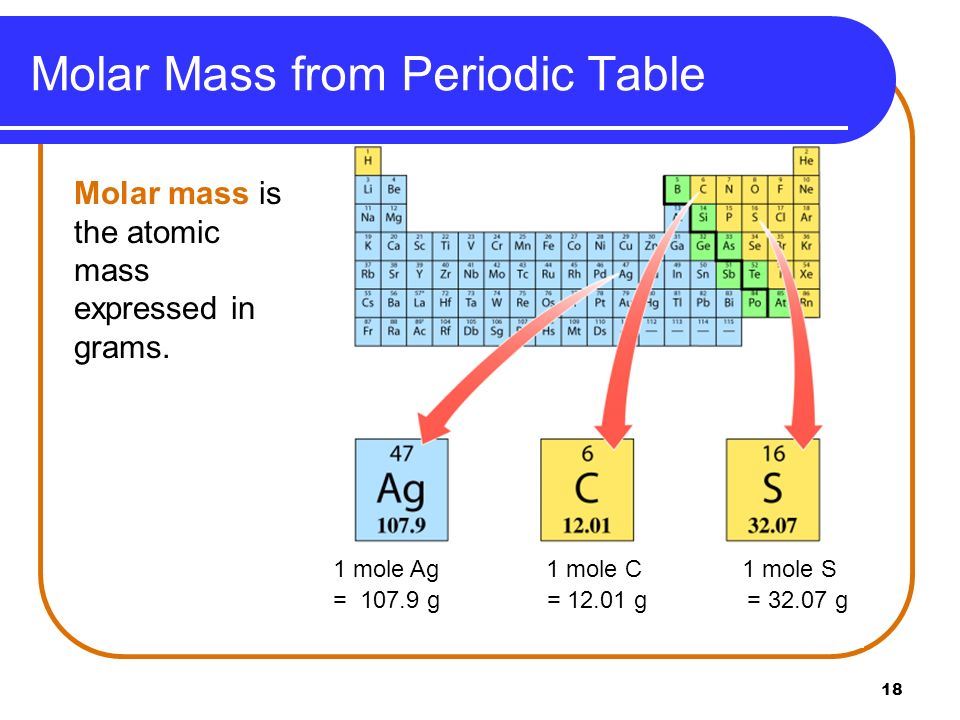
It has a boiling point of -252.8C and a melting point of -259.2 C. Mass numbers of typical isotopes of Hydrogen are 1 2. Hence the average atomic mass of hydrogen is equal to 1.00794 amu. Hydrogen has an atomic number of 1 and atomic mass of 1.007825g.mol-1. It has many uses in modern days such as glowing watches, fusion reactions, and self-powered devices. The percent composition of protium is 99.985. Protium consist of 1 electron and 1 proton, without any neutron, hence it has 1.007 amu as its atomic mass, while deuterium, has 1 electron, 1 proton, and 1 neutron, so it has a mass of 2. Note: Hydrogen has 3 isotopes, protium, deuterium and tritium. The total atomic weight of four atoms of hydrogen is calculated at 4.032. Hence, the atomic mass of hydrogen is 1.007 amu, which is 1.66 ×1024g 1.66 × 10 24 g. Tritium is produced in a very small amount by the reaction of cosmic rays with atmospheric gases and while testing a nuclear weapon. The atomic weights of hydrogen and helium are 1.008 and 4.003 respectively. the half-life of 12.31 years it decays into helium-3, which means it is radioactive. Protium ( H 1 ) \left( \right) ( H 3 )- It contains a proton and two neutrons in the nucleus. Another startup, Universal Hydrogen, is chasing the same goal of hydrogen-powered flight.To confirm your answer, compare the calculated value to the weighted mass displayed on the periodic table.There are three naturally occurring isotopes of hydrogen- Beside the Washington state booth at the Paris Air Show, Inslee announced the imminent groundbreaking of a truly green jet fuel production facility with a company that uses CO2 and water to make a. The atomic mass of oxygen (use percent natural abundance data from Table 2.5.1) would be calculated as follows:Ītomic mass = (%1)(mass 1) (%2)(mass 2) (%3)(mass 3) The molar mass of an element or molecule is the total mass in grams of all the atoms that comprise a mole of a certain. For chlorine, there are only two naturally occurring isotopes so there are only two terms.Ītomic mass = (%1)(mass 1) (%2)(mass 2) ⋯Īnother example: oxygen exists as a mixture that is 99.759% 16O, 0.037% 17O and 0.204% 18O. This is repeated until there is a term for each isotope. The weighted average is determined by multiplying the percent of natural abundance by the actual mass of the isotope. One atomic mass unit is equal to 1.66 x 10 -24 grams. The unit of measure for mass is the atomic mass unit (amu). Therefore this resulting atomic mass is calculated from naturally-occurring isotopes and their abundance. Each silicon atom is bonded to three hydrogen atoms (H), so there are a total of 9 hydrogen atoms in each. 
The atomic mass of an element is the weighted average of the atomic masses of the naturally occurring isotopes of that element. Note that each element may contain more isotopes. Trisilylamine Element Mass Percent Silicon 84. Why? The reason is that we need to take into account the natural abundance percentages of each isotope in order to calculate what is called the weighted average. = 35.968 \,u \nonumber \]Īs you can see, the average atomic mass given in the last column of the table above (35.453) is significantly lower.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
